Dosing & Administration
PIQRAY is a once-daily medication given in combination with fulvestrant1
Continue treatment until disease progression or until unacceptable toxicity occurs1
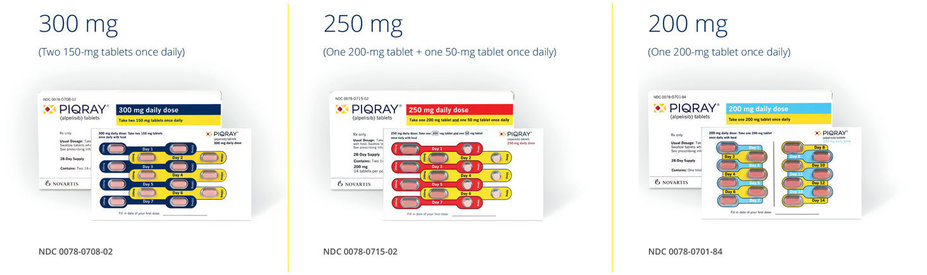
*Tablets should not be chewed, crushed, or split prior to swallowing. No tablet should be ingested if it is broken, cracked, or otherwise not intact.
Blister pack is designed to help patients stay on track with treatment1
PIQRAY approved doses:
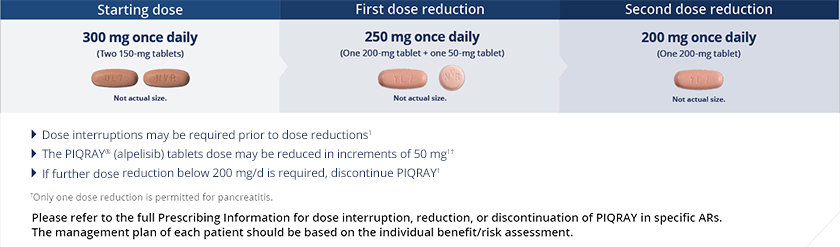
Dose modifications for PIQRAY
Certain ARs may require dose modifications1
ARs, adverse reactions.
Patient management resources

BROCHURE
Monitoring AR Checklist
Monitor for ARs before and during treatment.